Information on Marketing Authorisation Variations Based on Text Updates According to the Updated CCDS Document
SÚKL informs marketing authorisation holders of regulatory approach to the validation of Type II variations consisting in updates of product information texts based on the Company Core Data Sheet (CCDS).
The Company Core Data Sheet (CCDS) is a document developed by the marketing authorisation holder for the purposes of monitoring and consolidating implemented changes in the product information texts (SmPC, PIL and labelling). This document is based on the assessment of new literature and proprietary data and serves exclusively for internal company purposes. When submitting applications for marketing authorisation variations consisting in text changes derived from the CCDS, it is therefore always necessary to submit both the literature sources and data supporting the proposed text changes, as well as a consolidated assessment of these data, preferably in the form of an Addendum to the Clinical Overview or a Clinical Overview. All text changes must be duly explained both in the application form and in the document containing the consolidated assessment (e.g. whether they constitute formal/editorial changes, changes based on new data, or changes implemented in accordance with the QRD template, etc.).
For example, see the table below:
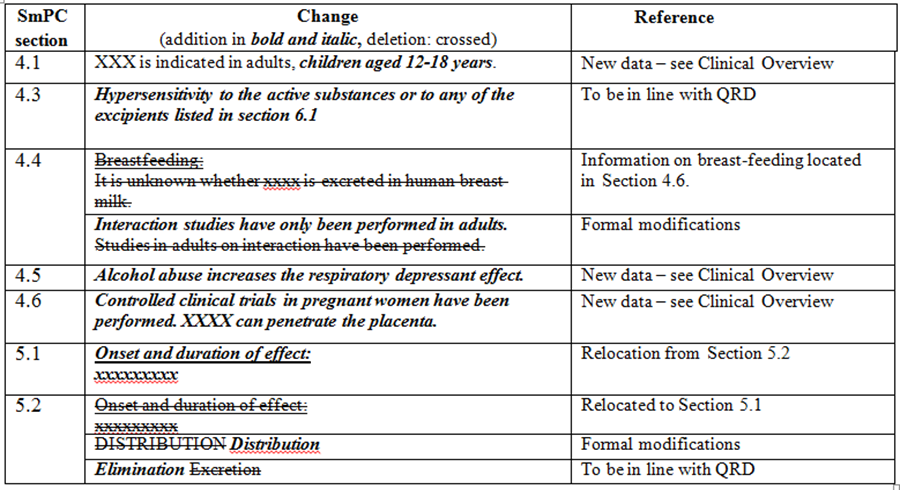
With regard to the classification of variations, as stated above, a variation application cannot be based solely on the CCDS document. For classification purposes, the underlying literature and proprietary data supporting the change must be taken as the basis. According to Question 4.17 of the CMDh document Questions and Answers on Variations, each individual reason for a variation to a medicinal product must be classified as a separate variation.
For marketing authorisation variations for medicinal products where the Czech Republic acts as RMS, or for nationally authorised medicinal products, the State Institute for Drug Control shall proceed as follows, depending on which parts of the dossier and SmPC are affected by the changes:
| SmPC Section | Dossier Part | Classification |
|---|---|---|
| Sections 2; 3; 6 | Pharmaceutical part of the dossier | Q – variations to the quality part of dossier |
| Sections 4.1 + 4.2 + 5 | Addition of a new indication associated with a change in posology | C.6 – a single classification |
| Sections 4.3 to 4.9 | Clinical Safety | C.4 – a single classification |
| Sections 4.6 to 5.3 | Preclinical Safety | C.4 – a single classification |
| Sections 5.1 + 5.2 | Clinical Efficacy | C.4 – a single classification (in case it is not related to the addition of a new indication) |
Variations classified as category “C” may be grouped within a single application (submitted as a grouping). Where the SmPC changes are interrelated, they may be submitted as a single variation (subject to appropriate justification). Where the changes are of a purely formal/editorial nature, they may be submitted within any category “C” variation of Type IB or Type II.
